Plastics Surface Energy Wetting Test Methods
By Scott Sabreen, The Sabreen Group
Question:
Our company uses corona discharge/air-plasma pretreatments before bonding to polyolefins. The product surface is textured with remote separation areas, so dyne testing is very difficult. Is there a way to test in-line production without using hazardous dyne solutions?
Answer:
Yes, there is a new breakthrough technology to measure contact angles in-line production without using chemical dyne solutions. The test is non-destructive, thus the part can be processed as usual (unlike dyne solution testing in which the part is scraped). Invented by Brighton Technologies Group, the Surface Analyst™ was engineered to put precision surface measurement technology in the hands of shop-floor technicians for operations where surface condition is critically important, such as: adhesive bonding, painting, coating, printing, decontamination, formulations, etc. Reference Picture 1, below.
breakthrough technology to measure contact angles in-line production without using chemical dyne solutions. The test is non-destructive, thus the part can be processed as usual (unlike dyne solution testing in which the part is scraped). Invented by Brighton Technologies Group, the Surface Analyst™ was engineered to put precision surface measurement technology in the hands of shop-floor technicians for operations where surface condition is critically important, such as: adhesive bonding, painting, coating, printing, decontamination, formulations, etc. Reference Picture 1, below.
The Surface Analyst™ uses contact angle (wetting measurement) to provide an immediate Go/No Go alert and a quantitative measure of the state of the surface. It operates in any orientation and works equally well on smooth or textured surfaces, such as grained TPO’s. Measurements are taken and logged in only 3 seconds. This data can be used for continuous process improvement, identifying quality drifts that may signal imminent problems, or to pinpoint the genesis of product failures.
The Surface Analyst™ was developed with funding from the US Air Force with the original goal of controlling cost and quality in aerospace applications involving adhesive bonding of advanced materials. The Surface Analyst™ serves a broad range of applications and industries using many different materials. Currently the largest customer base is automotive manufacturers concerned with paint adhesion on polymers such as TPO, and adhesive bonding. The measurement methodology was validated over several years in part by a series of studies correlating Surface Analyst™ measurements with bond failures on materials contaminated at controlled levels.
Presently, some of the world’s premier manufacturers are utilizing the Surface Analyst™ as their new standard protocol for measuring contact angles/surface energy (eliminating consumable dyne chemical solutions). Brighton Technologies Group will present this technology at the Decorating and Assembly Division of the Society of Plastics Engineers Topical Conference 2012 (June 5 and 6 in Indianapolis, IN).
Regarding surface wetting test methodologies, it’s helpful to generally understand basic principles of surface science. Based upon your application description (textured surface with isolated remote areas) you may want to consider using dyne bottled solutions (with cotton applicator swab) method, or Brighton Technologies Group’s Surface Analyst™ device and/or other procedures.
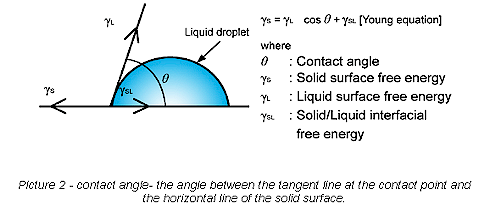
Plastic substrates are difficult to bond because they are “hydrophobic” (not naturally wettable), possess poor surface wettability (i.e., low surface energies), nonpolar-inert structures and possess poor surface-chemical functionality. The contact angle and its physical properties of interaction between solids and liquids provide valuable information in determining optimal adhesion bonding surface wettability conditions. The angle formed by the solid surface and the tangent line to the upper surface at the end point is called the contact angle; it is the angle between the tangent line at the contact point and the horizontal line of the solid surface. Reference Picture 2.
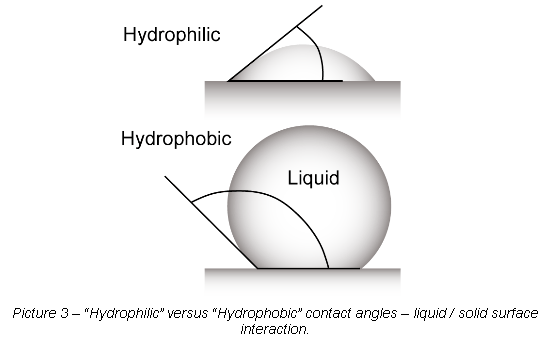
 The bubble/droplet shape is due to the molecular forces by which all liquids, through contraction of the surface, tend to form the contained volume into a shape having the least surface area. The intermolecular forces that contract the surface are termed “surface tension.” Surface tension, a measurement of surface energy, is expressed in “dynes/cm” (or mN/m SI units). The higher the surface energy of the solid substrate relative to the surface tension of a liquid (water, printing inks, adhesives/encapsulation, coatings, etc.), the better will be its “wettability,” and the smaller will be the contact angle. As a general rule, acceptable bonding adhesion is achieved when the surface energy of a substrate (measured in dynes/cm) is approximately 8 to 10 dynes/cm greater than the surface tension of the liquid. Robust “hydrophilic” surfaces need to be achieved versus non-wettable “hydrophobic” surfaces. Reference Picture 3.
The bubble/droplet shape is due to the molecular forces by which all liquids, through contraction of the surface, tend to form the contained volume into a shape having the least surface area. The intermolecular forces that contract the surface are termed “surface tension.” Surface tension, a measurement of surface energy, is expressed in “dynes/cm” (or mN/m SI units). The higher the surface energy of the solid substrate relative to the surface tension of a liquid (water, printing inks, adhesives/encapsulation, coatings, etc.), the better will be its “wettability,” and the smaller will be the contact angle. As a general rule, acceptable bonding adhesion is achieved when the surface energy of a substrate (measured in dynes/cm) is approximately 8 to 10 dynes/cm greater than the surface tension of the liquid. Robust “hydrophilic” surfaces need to be achieved versus non-wettable “hydrophobic” surfaces. Reference Picture 3.
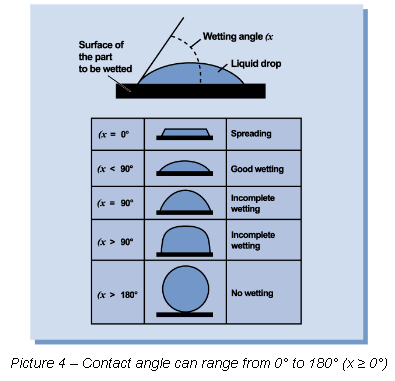
 Surface wetting tension can possess varying degrees of contact angles ranging from zero (0) to 180-degrees, as shown in Picture 4.
Surface wetting tension can possess varying degrees of contact angles ranging from zero (0) to 180-degrees, as shown in Picture 4.
Hydrophilic Surface – ©2012 The Sabreen Group, Inc.
Hydrophobic Surface – ©2012 The Sabreen Group, Inc.
Partial Surface Wetting – ©2012 The Sabreen Group, Inc.
Surface tension measurements can vary considerably by individual (technique) and the interpretation of the center liquid behavior. While dyne pen(s) kits may seem simple to use and economical they provide very basic Go-No/Go attribute data, not precise measurements of surface tension (contact angle) variable data (6-Sigma process control). These conditions can make it difficult for Manufacturing and QC personnel to determine whether or not the surface pretreatment has properly functionalized the treated surface(s). I advocate the method of dyne bottled solutions using a cotton applicator swab in accordance with ASTM D2578 test procedure.
©2012 The Sabreen Group, Inc. Photos and movie clips are the exclusive property of The Sabreen Group, Inc.

Very interesting article. This instrument should find widespread uses in manufacturing settings. This article “wetted” my interest in learning more about this instrument, such as how it works, capabilities, and limittions.
Bernie,
I’m posting your comment…it is the first one…I’m getting to learn how to do something new. Thank you for your kind feedback.
Scott Sabreen